Learning Objectives
- Outline nucleophiles
- Outline nucleophilic substitution reaction in terms of leaving group
- [AHL] Describe the SN1 and SN2 mechanisms
Part 1 Nucleophiles

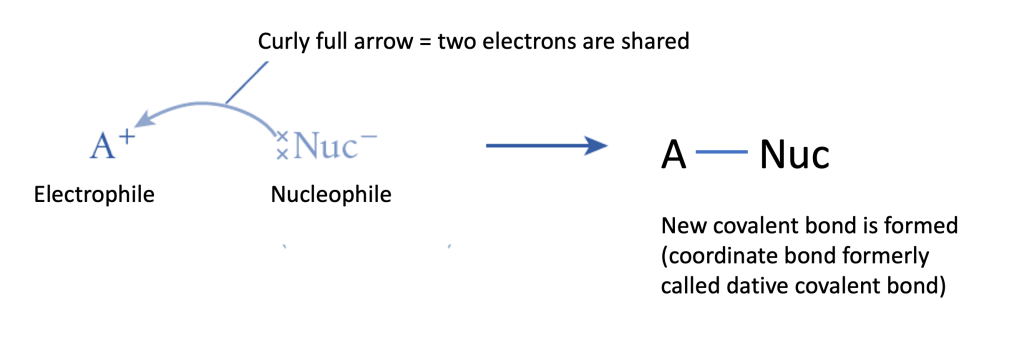
- Nucleophile (Nu⁻ or Nu:) – A species with a lone pair of electrons that attacks an electrophilic centre.
- Examples: OH⁻, CN⁻, NH₃, H₂O.
- Nucleophiles donate electron pairs to form coordination bonds
Part 2: Nucleophilic Substitution
Use the slideshow below to see the overview of the reaction:
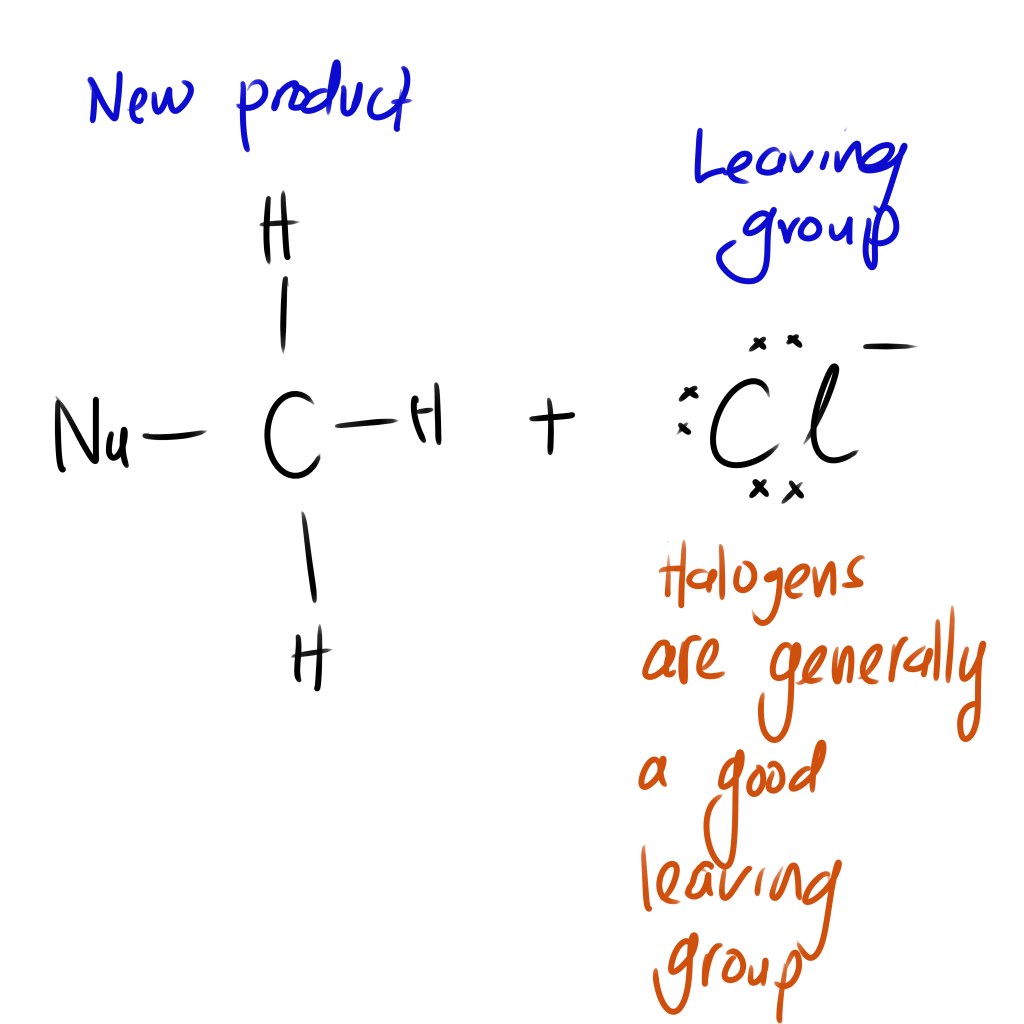
The result of this reaction:
- New product is formed
- Cl left the molecule –> is now called the leaving group
There are two types of nucleophilic substitution which is covered in AHL only:
- SN1 occurs on tertiary C
- SN2 occurs on primary C
Part 3: [AHL] SN2 Mechanism
- Aprotic solvents
- are not able to form hydrogen bonds as they do not contain –OH or –NH bonds,
- although they may have strong dipoles.
- they solvate the metal cation (e.g. Na+) rather than the nucleophile (OH–).
- The unsolvated bare nucleophile has a higher energy state and this increases the reaction rate
- Suitable solvents
- Propanone (CH3)2CO)
- Ethanenitrile (CH3CN)
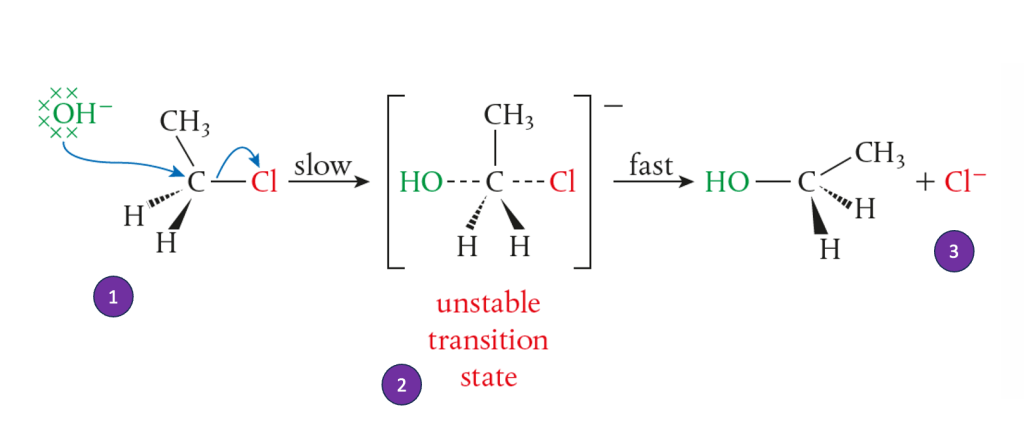
SN2 (Bimolecular Nucleophilic Substitution)
- Mechanism: Occurs in one step.
- From the above figure, here are the explanations
- The nucleophile attacks from the opposite side as the leaving group departs…
- …forming a transition state
- The unstable transition state returns to a stable state by losing Cl
- Reaction Rate: Depends on both the substrate and nucleophile concentrations
- Therefore the order bimolecular = second order

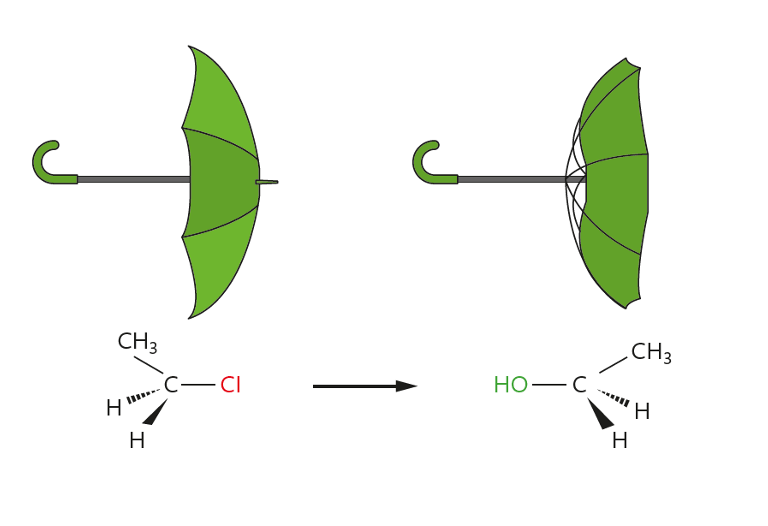
- Stereochemistry: Always leads to inversion of configuration (Walden inversion)
Part 4: [AHL] SN1 Mechanism
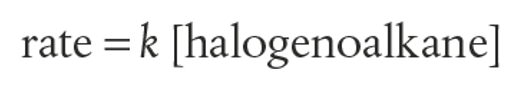
The overall rate expression is order 1 as the slow step is unimolecular (SN1)
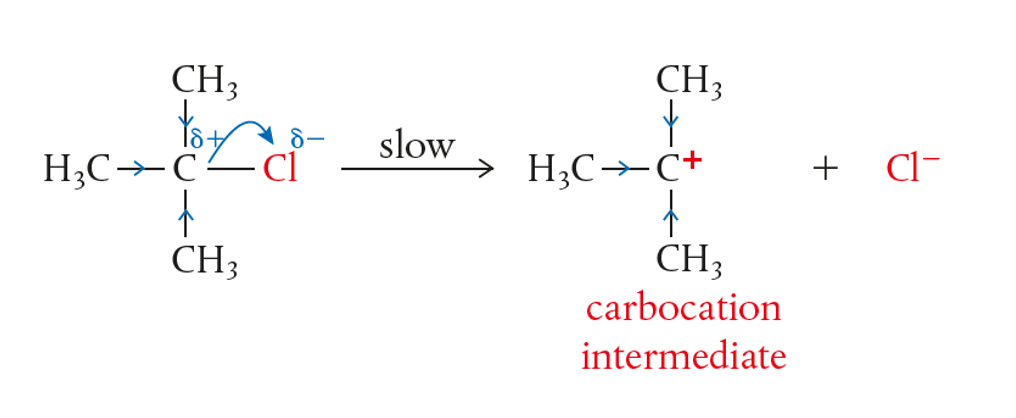
STEP 1: Formation of Carbocation (slow step)
- The central carbon of a tertiary haloalkane is protected by bulky alkyl groups. This makes it harder for other nucleophile to attack. This is called steric hindrance
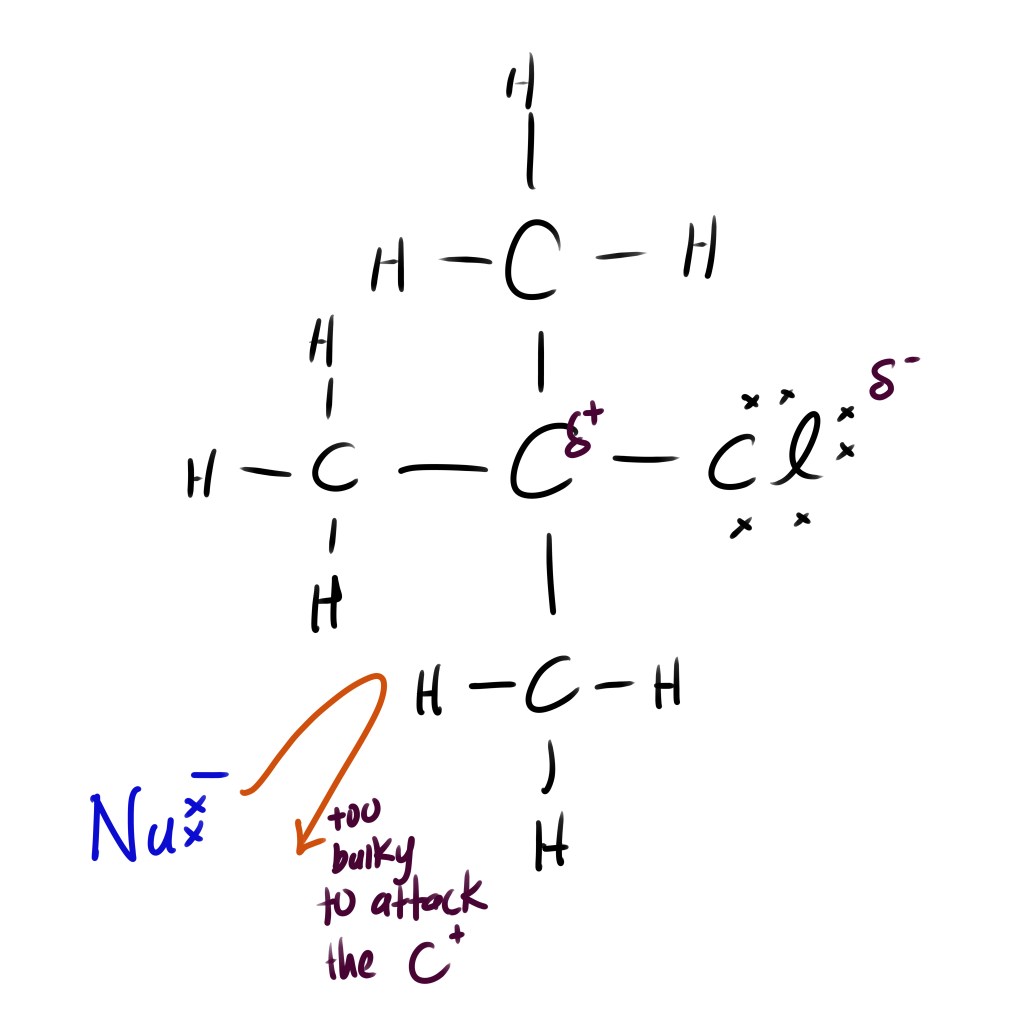
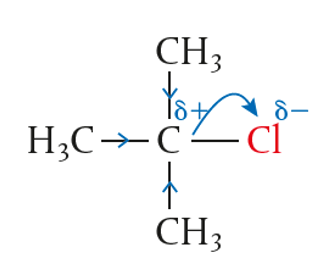
In contrast to primary haloalkane, the central carbon atom in tertiary haloalkane has other sources of electrons around it. Therefore, when the halogen such as Cl pulls the shared electron, the central carbon is stabilised by the surround alkyl groups
- A carbocation intermediate is produced.
- Therefore, it is important that the solvent helps in stabilising the intermediate
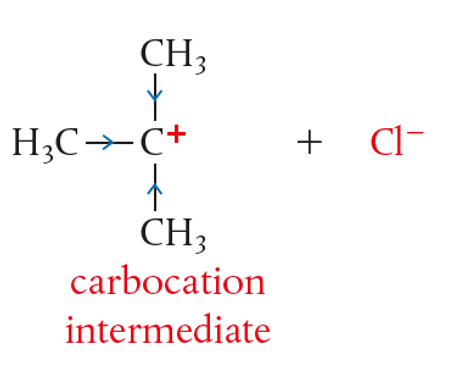
- Halide leaves forming an intermediate (stabilised by positive inductive)
- Polar and protic solvents contain –OH or –NH and so are able to form hydrogen bonds.
- stabilising the positively charged intermediate by solvation involving ion–dipole interactions.
- Suitable solvents = water, alcohols, and carboxylic acids
The formation of carbocation from the tertiary haloalkane is slow!
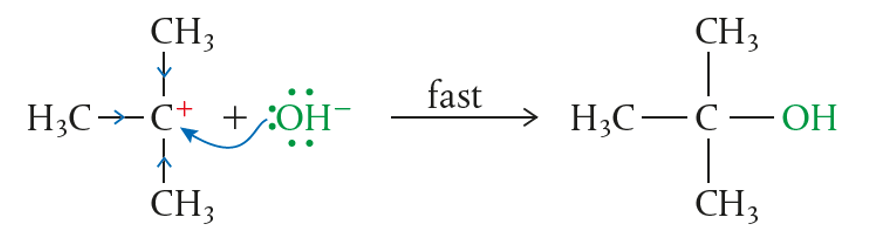
STEP 2:
- Carbocation has a planar shape
- This means that other Nu– can attack from any direction now
Part 5: [AHL] Overview of SN1 and SN2
| Feature | SN1 | SN2 |
|---|
| Mechanism | Two-step with carbocation formation | One-step with transition state |
| Rate Law | First-order (depends on RX) | Second-order (depends on RX and Nu) |
| Substrate | 3° > 2° (carbocation stability) | 1° > 2° (less steric hindrance) |
| Stereochemistry | Racemization | Inversion (Walden inversion i.e the umbrella) |
| Solvent | Polar protic (H₂O, alcohols) | Polar aprotic (DMSO, acetone) |
| Nucleophile | Weak (H₂O, ROH) | Strong (OH⁻, CN⁻, I⁻) |
Secondary halogenoalkanes
It is not possible to be precise about the mechanism of nucleophilic substitution in secondary halogenoalkanes, as data show that they usually undergo a mixture of both SN1 and SN2 mechanisms.
Part 6: [AHL] The influence of the leaving group (halogen)
The strength of the carbon–halogen bond (More important factor)
Ease to break:
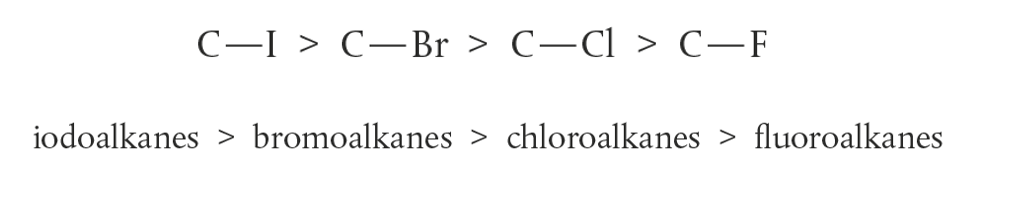
C-I has the lowest bond enthalpy where easier for I– to leave

![ESS 8.1.3 [AHL] Biocapacity and Environmental Migration](https://mypytrclass.com/wp-content/uploads/2025/12/image-4.png?w=1024)




![ESS 8.3.4 [AHL] Photochemical Smogs and Tropospheric Ozone](https://mypytrclass.com/wp-content/uploads/2025/12/image-1.png?w=1024)
