Author: Peter M.K.H JAMAL
-

Biology A1.1.1 Water
Learning Objectives Part 1 Theory of Water Hydrogen Bonding in Water Due to the high electronegativity of O, it pulls the shared electrons towards it making H atoms to be an area of electron deficit (δ+). The O side as the result is now δ– as it is an electron rich region When two water…
-

ESS 3.1.1 Defining Biodiversity
Learning Objectives Part 1: Biodiversity Definition Definition of Biodiversity Diversity is a measure of both the number of species in an area and their relative abundance. Biodiversity is the total diversity of living systems and exists at several levels. It is a result of habitat diversity, species diversity and genetic diversity Mt. Kinabalu is located in…
-

Chemistry R3.3.5 Electron Sharing in Lewis Structures
Learning Objectives Part 1: Interactions Between Lewis Acids and Lewis Bases The Lewis acid-base theory describes the interaction between electron pair donors (Lewis bases) and electron pair acceptors (Lewis acids). This theory is broader than the Bronsted-Lowry definition and applies to a wider range of reactions, including coordination complexes and organic reactions. Terminologies Lewis Acid-Base…
-
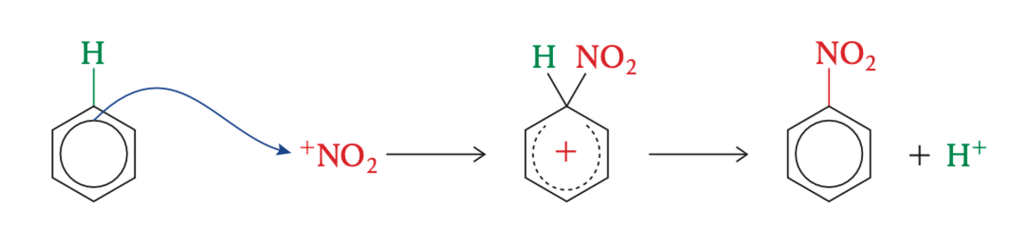
Chemistry R3.3.3 Electrophilic Substitution
Learning Objectives Part 1 Electrophilic Substitution Watch the video to find out more Step 1: Formation of nitronium ion Step 2: Electrophilic substitution
-
Chemistry R3.3.1 Fissions and Radicals
Learning Objectives Part 1: Heterolytic and Homolytic Fission Homolytic Fissions Homolytic fission is the breaking of a covalent bond in which each atom takes one electron from the bond, forming two free radicals. Heterolytic Fissions Heterolytic fission is the process where a covalent bond breaks unevenly, resulting in one atom receiving both electrons from the…
-
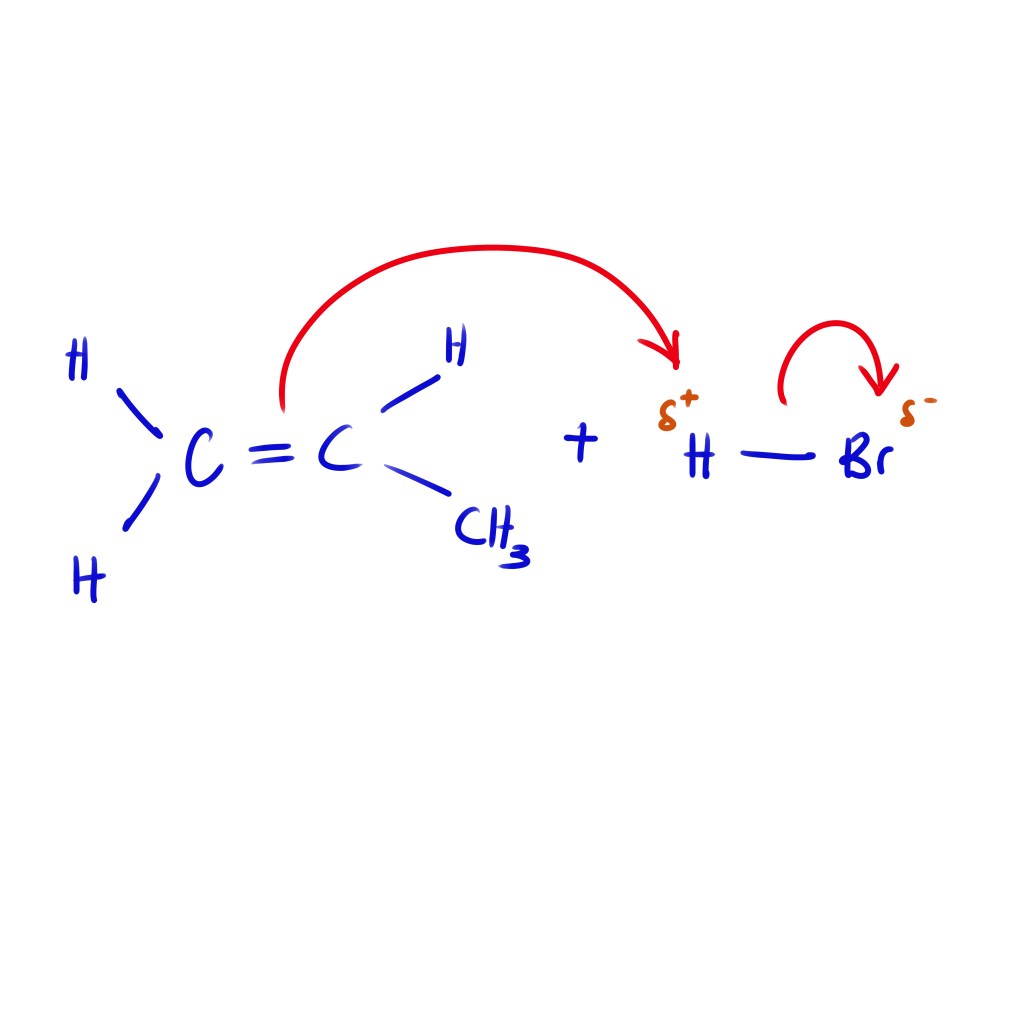
Chemistry R3.3.2 Electrophilic Additions
Learning Objectives Part 1 What are Electrophiles? Definition: Part 2: Electrophilic Addition Reactions Introduction to alkenes In general, electrophilic addition requires the electrophile to undergo a heterolytic fission. This type of split is uneven where one side will get both of the shared electrons (an electron pair). This leaves one part to become a cation…
-
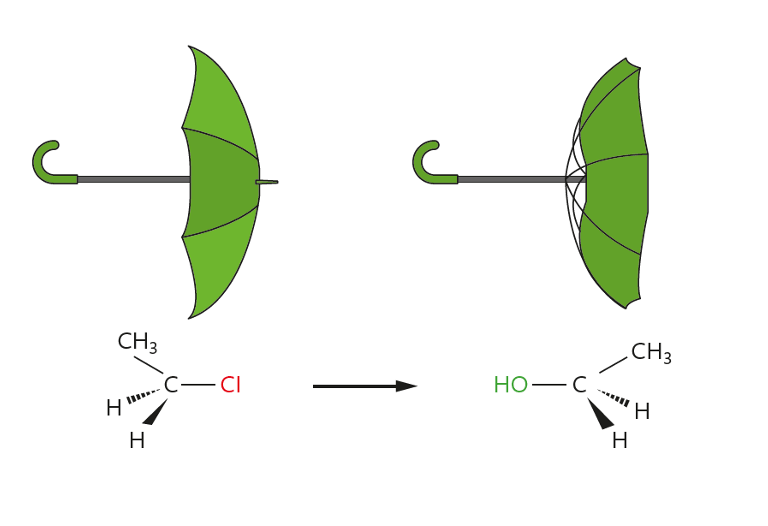
Chemistry R3.3.4 Nucleophilic Substitutions
Learning Objectives Part 1 Nucleophiles Part 2: Nucleophilic Substitution Use the slideshow below to see the overview of the reaction: The result of this reaction: There are two types of nucleophilic substitution which is covered in AHL only: Part 3: [AHL] SN2 Mechanism SN2 (Bimolecular Nucleophilic Substitution) Part 4: [AHL] SN1 Mechanism The overall rate…
-

Chemistry S1.5 Limiting Reactants
Learning Objectives Part 1: Limiting Reactant In every reaction involving two reactants, one may become limiting and the other will be in excess. This means that… Based on this graph, oxygen is the limiting reactant as it is completely used to form water. There are some amount of hydrogen gas remained unreacted at the end…
-

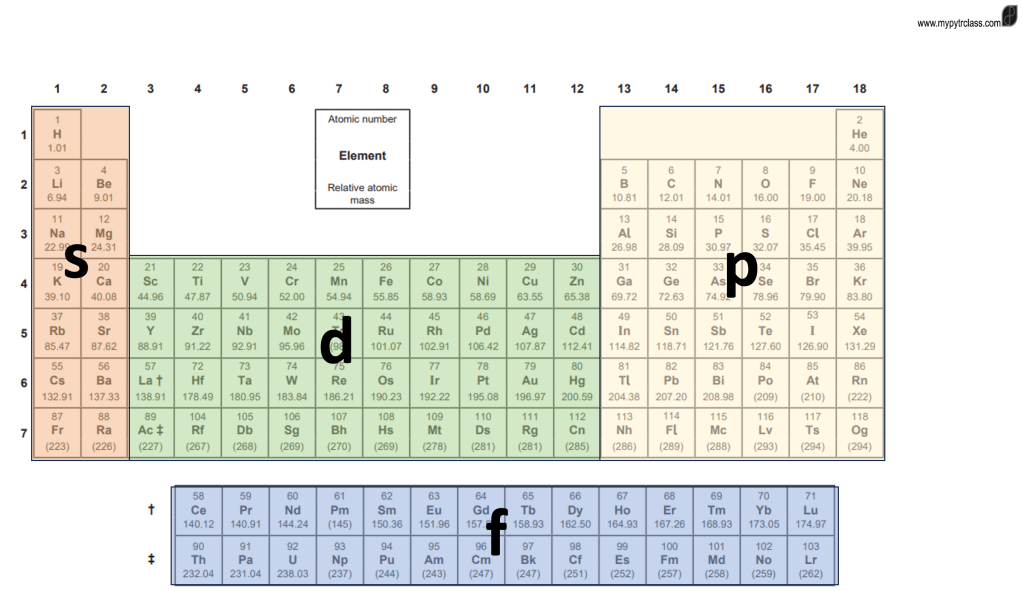
Chemistry S1.2 Nuclear Atom
Learning Objectives Part 1: Atomic Model An atom has 3 groups subatomic particles: When writing an ion, the A and Z do not change from its element. The number electron(s) that the atom has received or lost is indicated at P Example: To sum: Atomic Number (Z) and Mass Number (A) Formula:A = Z +…
-

Chemistry S1.4 The Mole Concept
Learning Objectives This section focuses on: Part 1: Relative Atomic Mass Relative Atomic Mass (Ar) is the weighted average mass of an atom of an element compared to 1/12th the mass of a carbon-12 atom. It takes into account the different isotopes of the element and their abundances. From the above, it is noted that…
-
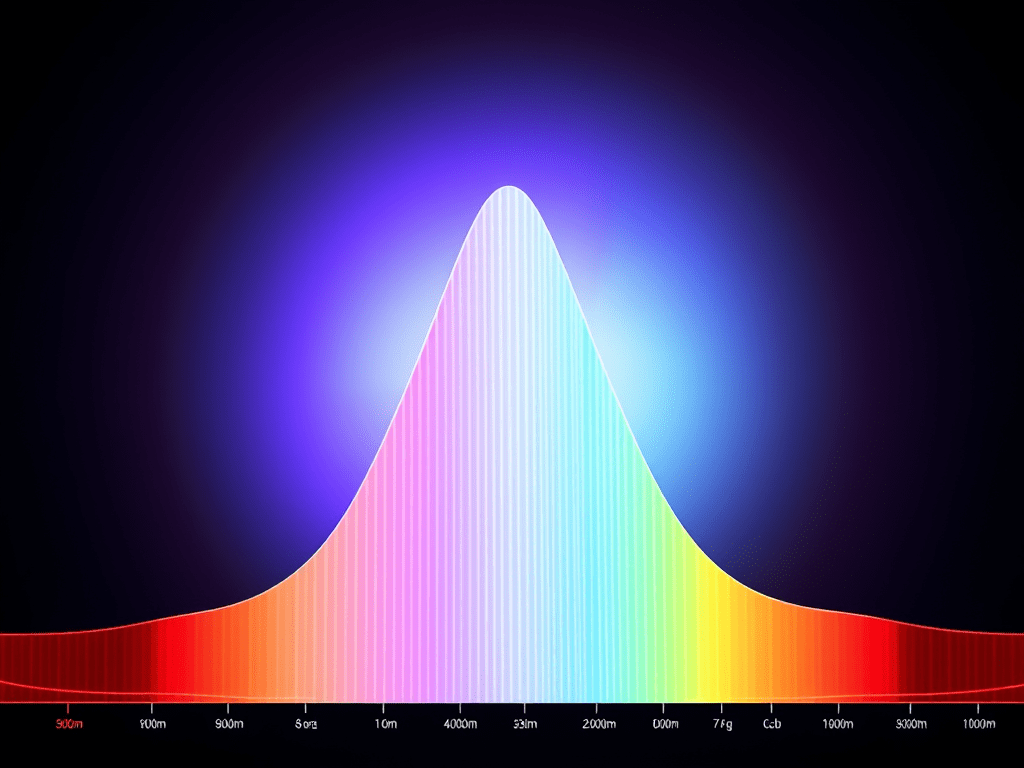
Chemistry S1.3 Electron Configuration
Learning Objectives Part 1: Absorption and Emission Spectra Did you know that the colour of flame correspond to a certain electromagnetic radiation? When burned, electrons are excited and when returned to the original energy level, photon is released. This photon may be visible light, infra-red or UV etc. The above are flame test of Na,…
-

Chemistry S1.1 Particulate Nature of Matters
Learning Objectives Part 1: Elements, Compounds and Mixtures All matters can be found in two major compositions: Part 2: Separation Techniques Part 3: States of Matter Kinetic Molecular Theory Property Solids Liquids Gases Distance between particles Close together Close but further apart than in solids Far apart Arrangement Regular Random Random Shape Fixed shape No…
-
![ESS 2.5.2 [Case Study] Primary and Secondary Succession](https://mypytrclass.com/wp-content/uploads/2025/02/image-12.png?w=1024)
ESS 2.5.2 [Case Study] Primary and Secondary Succession
Learning Objectives Part 1: Primary Succession Case Study: Primary Succession on Krakatau, Indonesia Part 2: Secondary Succession Case Study: Secondary succession in the Broadbalk Wilderness, Rothamsted, UK Part 3: Pioneer Vs Climax Communities Part 4: Summary of Changes Changes Over Time During Succession Succession is the natural process of change in the species composition and…
-
ESS 2.5.1 Zonation and Succession
Learning Objectives Zonation “Zonation describes the variation in biological communities along an environmental gradient” On rocky shores, distinct zones extend from the lower to the upper shore, each characterised by specific plant and animal distributions. Example: Seaweeds, in particular, exhibit clear zonation patterns Using Transect to Investigate Zonation Succession Ecosystems are dynamic systems that undergo…