Author: Peter M.K.H JAMAL
-
![Biology B1.2.2 [AHL] Proteins Structures](https://mypytrclass.com/wp-content/uploads/2025/02/screenshot-2025-02-25-at-14.30.09.png?w=1024)
Biology B1.2.2 [AHL] Proteins Structures
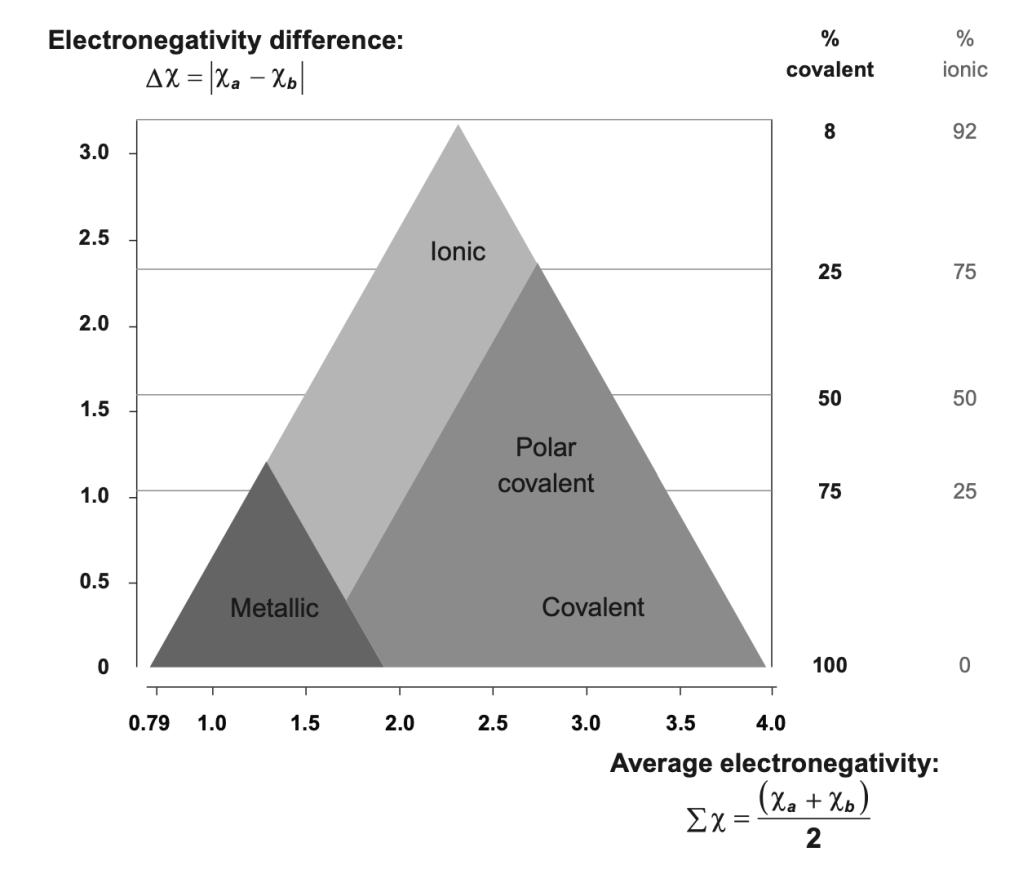
Learning Objectives Part 1: The R Group of Amino Acids The 20 amino acids used by ribosomes to form polypeptides vary significantly in the chemical nature of their R-groups. When amino acids link into a polypeptide, their amine and carboxyl groups form peptide bonds, leaving a free amine group (–NH₂) at one end and a…
-
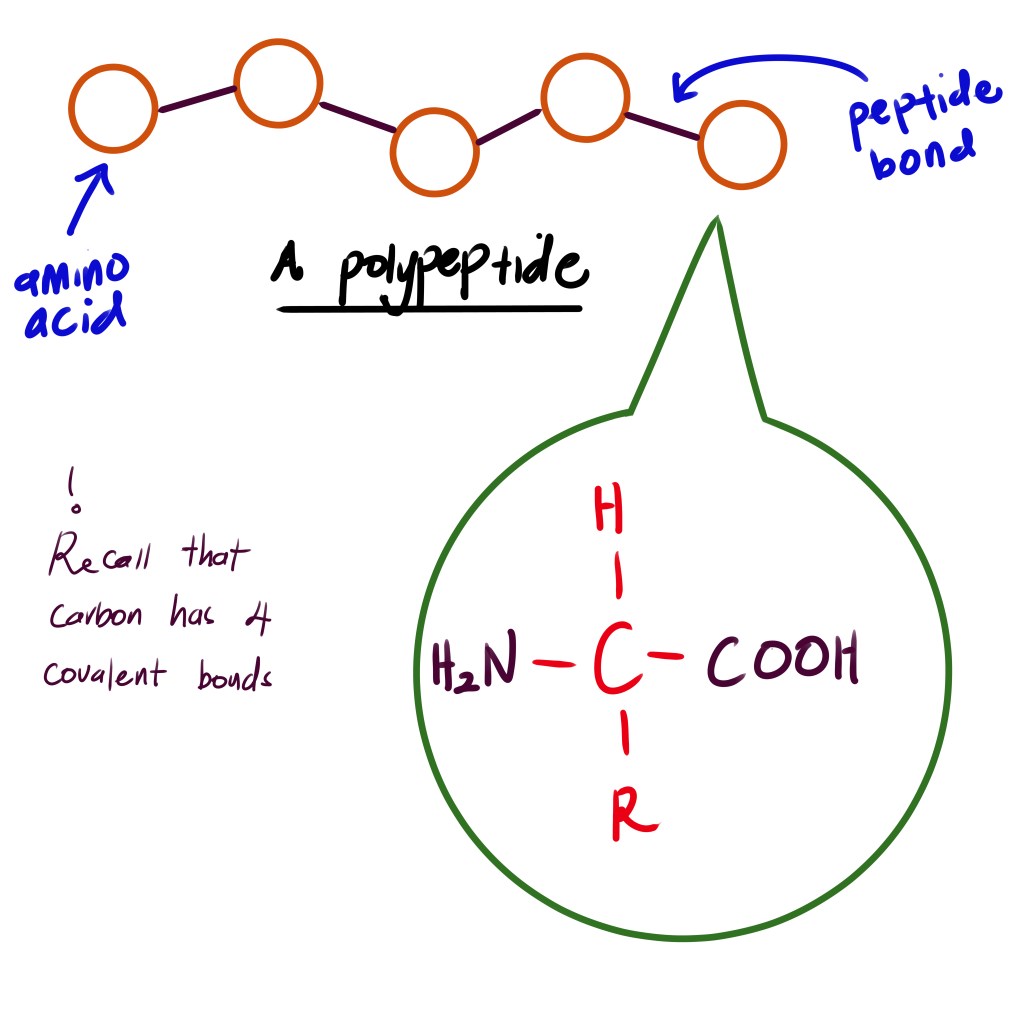
Biology B1.2.1 Proteins
Learning Objectives Abstract:The diversity in protein form and function is fundamentally linked to the amino acid sequence of their polypeptides. The 20 chemically diverse amino acids can be arranged in countless sequences, analogous to letters forming words, though only a fraction of possible sequences occur in nature. The specific sequence of amino acids determines a…
-

Biology B1.1.3 Lipids and Phospholipids
Learning Objectives Part 1: Hydrophobic Properties Properties and Types Lipids are a diverse group of substances found in living organisms. They dissolve in non-polar solvents such as ethanol, toluene, and propanone (acetone). They are sparingly soluble in water-based (aqueous) solvents, making them hydrophobic. However, lipids are not repelled by water; they are simply more attracted…
-

Biology B1.1.2 Carbohydrates
Learning Objectives Part 1: Structures Roles of Monosaccharides There are two type of glucose: alpha and beta. When becomes polymer, they form different structures Part 2: Polysaccharides 1. Polysaccharides as Energy Storage: Advantages of Starch & Glycogen: 2. Cellulose Part 3: Glycoproteins Role of Glycoproteins in Cell–Cell Recognition ABO Glycoproteins & Blood Transfusion
-
Biology B1.1.1 Biological Macromolecules
Learning Objectives Part 1: Roles of Carbon in Biological Systems Part 2: Summary of macromolecules Part 3: Condensation and Hydrolysis The making of large polymers often requires condensation reaction. The breaking down of the large polymers would be the reverse of condensation reaction and this is called hydrolysis. Both ways usually require enzymes or other…
-
![Biology A1.2.4 [AHL] Further About DNA](https://mypytrclass.com/wp-content/uploads/2025/02/jpeg-image-4c3a-9fb3-a8-0.jpeg?w=1024)
Biology A1.2.4 [AHL] Further About DNA
Learning Objectives Part 1: Directionality of RNA and DNA Directionality of RNA and DNA in Enzymatic Processes Replication Transcription Translation Part 2: Maintaining DNA Helical Structure Purine-pyrimidine Chargaff’s rules Part 3: What are Nucleosomes? DNA is packed (condensed) in the nucleus in a form of chromosomes. To do this, the DNA must wrap around many…
-

Biology A1.2.3 DNA as Universal Genetic Materials
Learning Objectives Part 1: Importance of Base Complimentary Role of Complementary Base Pairing in DNA Replication and Gene Expression DNA Replication Gene Expression Part 2: DNA as Universal Genetic Materials Diversity of DNA Base Sequences and Limitless Information Storage Conservation of the Genetic Code and Universal Common Ancestry
-
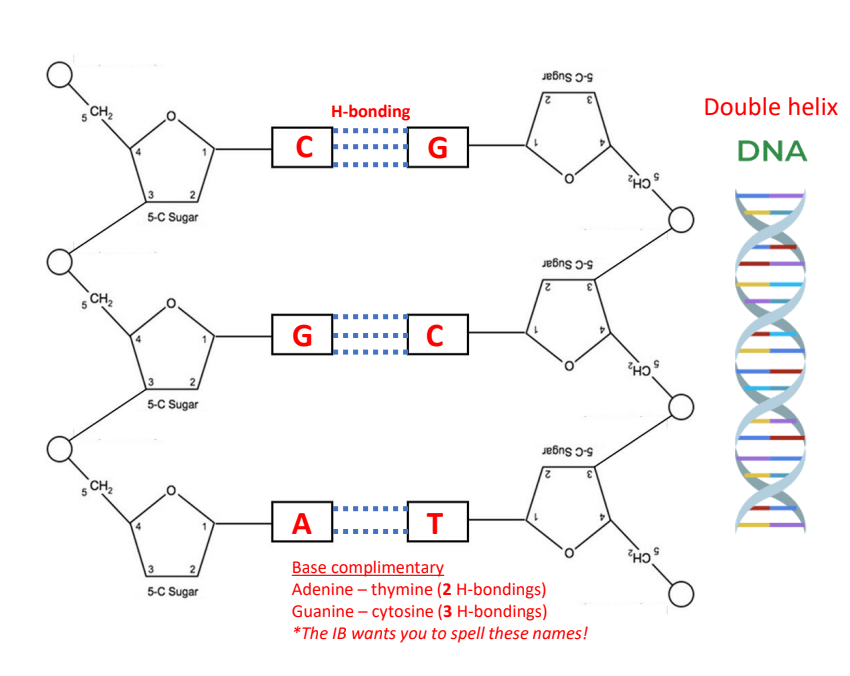
Biology A1.2.2 Structures of RNA and DNA
Learning Objectives Part 1: Condensation of Nucleotides Part 2: Structure and Roles of RNA Roles of RNA Part 3: Structure of DNA Structure of DNA as a Double Helix Part 4: Differences of DNA and RNA Ribose sugar is the sugar of RNA Deoxyribose sugar is the sugar of DNA
-

Biology A1.2.1 Nucleotides Make Nucleic Acid
Learning Objectives Part 1: Roles and Features of Nucleic Acids DNA as genetic materials for all living organisms Types of nucleic acids What are nucleotides? Part 2: DNA Backbone and Genetic Bases When there are many nucleotides linked together, they will look like a long strand of thread with the nitrogenous bases sticking out. This…
-

Biology A1.1.2 Water on Earth and Beyond
Learning Objectives Part 1: Origin of Earth’s Water Earth’s Water Hypothesis of Earth’s Water Origin Part 2: Factors of Earth’s Water Retention Two key factors contributed to Earth’s ability to retain water after its delivery by asteroids: Part 3: Goldilocks Zone In the fairy tale Goldilocks and the Three Bears, a young girl tries three…
![Chemistry S3.1.6 [AHL] Transition Metals](https://mypytrclass.com/wp-content/uploads/2025/02/screenshot-2025-02-27-at-21.32.24.png?w=1024)